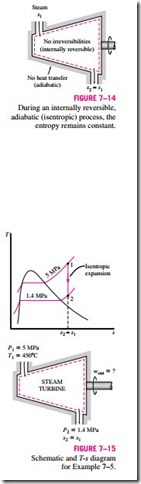
■ ISENTROPIC PROCESSES
We mentioned earlier that the entropy of a fixed mass can be changed by (1) heat transfer and (2) irreversibilities. Then it follows that the entropy of a fixed mass will not change during a process that is internally reversible and adiabatic (Fig. 7–14). A process during which the entropy remains constant is called an isentropic process. It is characterized by
That is, a substance will have the same entropy value at the end of the process as it does at the beginning if the process is carried out in an isentropic manner.
Many engineering systems or devices such as pumps, turbines, nozzles, and diffusers are essentially adiabatic in their operation, and they perform best when the irreversibilities, such as the friction associated with the process, are minimized. Therefore, an isentropic process can serve as an appropriate model for actual processes. Also, isentropic processes enable us to define efficiencies for processes to compare the actual performance of these devices to the performance under idealized conditions.
It should be recognized that a reversible adiabatic process is necessarily isentropic (s2 = s1), but an isentropic process is not necessarily a reversible adiabatic process. (The entropy increase of a substance during a process as a result of irreversibilities may be offset by a decrease in entropy as a result of heat losses, for example.) However, the term isentropic process is customarily used in thermodynamics to imply an internally reversible, adiabatic process.