Semiconductors are the basic components of electronic equipment. The more commonly used semiconductors are the diode (used to rectify),
the transistor (used to amplify), and the integrated circuit (used to switch or amplify). The primary function of semiconductor devices is to control voltage or cur- rent for some desired result.
Advantages of semiconductors include the following:
● Small size and weight
● Low power consumption at low voltages
● High efficiency
● Great reliability
● Ability to operate in hazardous environments
● Instant operation when power is applied
● Economic mass production
Disadvantages of semiconductors include these:
● Great susceptibility to changes in temperature
● Extra components required for stabilization
● Easily damaged (by exceeding power limits or reversing polarity of operating voltage, and by excess heat when soldering into a circuit)
Semiconduction in Germanium and Silicon
Semiconductor materials have characteristics that fall between those of insulators and conductors. Three pure semiconductor elements are carbon (C), germanium (Ge), and silicon (Si). Those suitable for electronic applications are germanium and silicon.
Germanium is a brittle, grayish-white element discovered in 1886. A powder, germanium dioxide, is recovered from the ashes of certain types of coal. The powder is then reduced to the solid form of pure germanium.
Silicon was discovered in 1823. It is found extensively in the earth’s crust as a white or sometimes colorless compound, silicon dioxide. Silicon dioxide (silica) can be found abundantly in sand, quartz, agate, and flint. It is then chemically reduced to pure silicon in a solid form. Silicon is the most commonly used semiconductor material.
Once the pure, or intrinsic material is available, it must be modified to produce the qualities necessary for semiconductor devices.
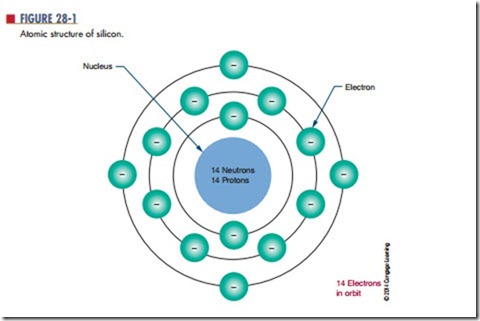
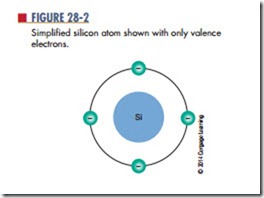
As described in Chapter 10, the center of the atom is the nucleus, which contains protons and neutrons. The protons have a positive charge and the neutrons have no charge. Electrons orbit around the nucleus and have a negative charge. Figure 28-1 shows the structure of the silicon atom. The first orbit contains two electrons; the second orbit contains eight electrons; and the outer orbit, or valence shell, contains four electrons. Valence is an indication of the atom’s ability to gain or lose electrons and determines the electrical and chemical properties of the atom. Figure 28-2
shows a simplified drawing of the silicon atom, with only four electrons in the valence shell.
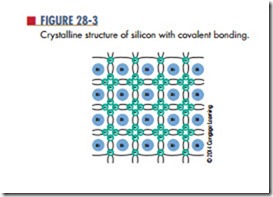
Materials that need electrons to complete their valence shell are not stable and are referred to as active materials. To gain stability, an active material must acquire electrons in its valence shell. Silicon atoms are able to share their valence electrons with other silicon atoms in a process called covalent bonding (Figure 28-3). Covalent bonding is the process of sharing valence electrons, resulting in the formation of crystals.
Each atom in such a crystalline structure has four of its own electrons and four shared electrons from four other atoms—a total of eight valence electrons. This covalent bond cannot support electrical activity because of its stability.
At room temperature, pure silicon crystals are poor conductors and behave like insulators. If heat energy is applied to the crystals, however, some of the electrons absorb the energy and move to a higher orbit, break- ing the covalent bond and allowing the crystals to sup- port current flow.
Silicon, like other semiconductor materials, is said to have a negative temperature coefficient, because as the temperature increases, its resistance decreases. The resistance is cut in half for every 6 degrees Celsius of rise in temperature.
Like silicon, germanium has four electrons in its valence shell and can form a crystalline structure. Germanium’s resistance is cut in half for every 10 degrees Celsius of temperature rise. Thus germanium appears to be more stable with respect to temperature change than silicon. However, germanium requires less heat energy to dislodge its electrons than does silicon. Silicon has a thousand times more resistance than germanium at room temperature.
Heat is a potential source of trouble for semiconductors and is not easy to control. Good circuit design minimizes heat changes. Its resistance is what makes silicon preferable to germanium in most circuits. In some applications, heat-sensitive devices are necessary. In these applications, the germanium temperature coefficient can be an advantage; therefore, germanium is used.
All early transistors were made of germanium. The first silicon transistor was not made until 1954. Today, silicon is used for most solid-state applications.
Questions
1. What is a semiconductor material?
2. Define the following terms:
a. covalent bonding
b. Negative temperature coefficient
3. Why are silicon and germanium considered semiconductor materials?
4. Why is silicon preferred over germanium?
5. When using semiconductors, what is the major problem that must be controlled?