SALINITY GRADIENT POWER GENERATION
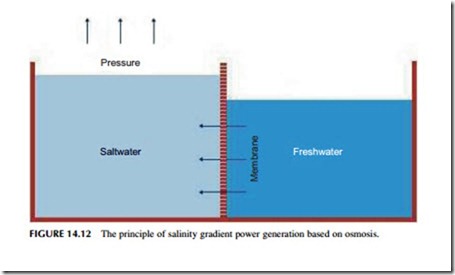
Salinity gradient power generation depends on the natural process of diffusion, or mixing. To understand how this can be used to generate electrical power, imagine a special box, divided into two compartments, as shown in Figure 14.12. One of the compartments is filled with salt water and the other with fresh water. If the division between the two compartments were carefully removed, without creating any turbulence, within a short period of time the water in both compartments would be salty. Diffusion would have mixed the fresh and salt water.
If the division between the two compartments is now replaced by a special, semi- permeable membrane that will allow water to pass through it but will not allow any
dissolved salt to cross, when one side is filled with salt water from the sea and the other side with fresh water, water will pass through the membrane from the fresh side to the salt side to try and equalize the concentration of salt in each chamber. Since this is not possible, in theory the water will continue to cross until a head of water builds up on the salt side that can create a pressure sufficient to prevent further water crossing. For seawater, this equilibrium would require a head of 240 m.
Osmotic Power
The pressure that can be generated as a result of diffusion is clearly significant. In principle, it should be possible to exploit this pressure to generate electric power. This is the idea behind the osmotic power plant, first proposed by Sidney Loeb during the 1970s.
In an osmotic power plant a cell is created containing pressurized seawater and this is opened, through a semi-permeable membrane, to fresh water. Fresh water will then diffuse by osmosis into the pressurized chamber increasing the pressure further. The excess pressure is then released by allowing some of the salt water to flow out through a turbine.
The development of osmotic power has been taken up by the Norwegian utility Statkraft that built a prototype plant that began operating in 2009. The plant has a generating capacity of 2–4 kW, although the company predicts this can be raised to 10 kW with better-performing membranes.
Statkraft is proposing to build a much larger plant in Norway to demonstrate the principle. The proposed size is 1–2 MW. The company has estimated that the global potential for osmotic power generation is around 1600 TWh annually.
Plants exploiting this technology would be sited at places where fresh water flows into the sea and would harness the mixing to generate electricity.
Vapor Compression
The vapor pressure of water that is generated above fresh water and salt water, if both are at the same temperature, is slightly different. Although the pressure difference is not great, it is possible to generate a vapor pressure from fresh water, under partial vacuum conditions, and then condense the vapor in salt water. This will create a pressure gradient that can be used to drive a gas turbine.
The pressure difference available is very small and would require a very large turbine, similar to an open cycle OTEC plant. This system does away with the need for a semi-permeable membrane but may be more difficult to develop than osmotic power.
Hydrocratic Power
If fresh water is introduced into the bottom of a vertical tube, pierced with holes, that is situated in salt water, then the salt water will force its way into the tube— driven by diffusion—to dilute the fresh water. The flow of water upwards through the tube is thus reinforced and a turbine placed in the flow can apparently generate more energy than is required to pump the fresh water into the pipe in the first place.
The process, christened hydrocratic power, has been patented and tests of its validity have been carried out. The scheme might be installed where power plant cooling water is discharged.
Reverse Electrodialysis
In principle, fresh and salt water can be used at two electrodes of a specially designed battery to produce an electric current directly. To achieve this, a series of cells are created with each separated from the next by a semi-permeable membrane. In this case, one membrane will only allow dissolved sodium ions to pass through it while the next will only allow dissolved chloride ions. Salt water is pumped into the first cell, fresh water into the second, salt water into the third, fresh water into the fourth, and so on along the line. Sodium ions will then diffuse from the salt water on one side into each fresh water cell, while chloride ions will diffuse from the other side. This will create a voltage separation between the saltwater cells on either side of the freshwater cell.
This process, called electrodialysis, was observed by R. Platte in 1954. The concept is the basis for the development of a power generation system by Netherland’s company REAPower.
COST OF MARINE POWER GENERATION
All the marine power generation technologies in this chapter are at an early stage of development of commercialization and realistic installation costs are difficult to establish. Based on recent estimates, the most expensive is a floating OTEC plant that has an estimated cost between $4200/kW and $12,300/kW.1 Wave power plants are somewhat less expensive with an installation cost between $4100/kW and $6300/kW. Tidal stream plants have an estimated cost between $1700/kW and $4000/kW. Salinity gradient technologies are too new for any reliable estimates of installed cost to be available.
While all these technologies appear relatively expensive to install, the energy has no cost and so the electricity they produce should be somewhat cheaper than these installation costs indicate. Wave power–generated electric- ity appears to be the most expensive today with an estimated cost of more than$180/MWh. For OTEC, the cost estimates are $70–220/MWh and for tidal stream they are $40–120/MWh. Cost estimates for osmotic pressure power are $70–130/MWh.