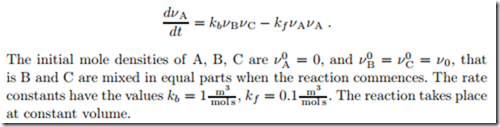Problems
Velocity of Reactions
Consider a reaction of the type A 1→ B + C which occurs according to dnA =−k nA (decay of A). Compute the mole density of A as a function of time, when the initial mole density of A is n0 , and neither B nor C are present initially. Assume constant volume.
Velocity of Reactions
Consider a reaction of the type B + C 0:: 2A which occurs according to
1. Derive the law of mass action for this reaction, and compute the equilibrium mole densities.
2. Compute the mole density of A as a function of time, and also compute the mole densities of B and C as functions of time.
3. Plot νA and νB as functions of time.
Related posts:
Transient and Inhomogeneous Processes in Open Systems:Counter-Flow Heat Exchangers
DUCTWORK SIZING:REMOTE SYSTEMS
Axial-Flow Turbines,Mean-Line Analysis and Design:Turbine stage design parameters.
Oil Furnaces:Waste Oil Furnaces
Mixtures:Ideal Mixtures
REFRIGERATION EQUIPMENT AND PROCESSES:WORKING WITH WATER-COOLING PROBLEMS
System control valves:Crankcase pressure regulator
Water Heaters:Lighting and Operating Instructions
Electrical Heating Systems - Crawl Space Exterior Walls